
Copyright Ó 1994, 1998, 2002, 2004 by Brian Fraser
All Rights Reserved; Rev 2004k
Virtually everyone who is scientifically literate is sure that the atom has a nucleus. The nuclear model of the atom has been in use by the scientific community and the textbook writers for about eighty years now. There are all sorts of "versions" of this model, but few scientists or science teachers would believe that there is anything seriously or obviously wrong with it or that it is not substantially in accord with proven fact. Nuclear physics is regarded as "hard" science and has been exempt from the type of debates that pester other scientific dogma such as human evolution. But suppose you encountered evidence that the atom does not in fact have a nucleus? How would you share your findings? If you wrote an article about the topic, do you think anyone would even bother to read it? If you gave a speech, would anyone listen to you? When your colleagues tried to convince you that you are surely out of your mind, that you are placing your career in jeopardy, . . .what would you do?
The Origin of the Nuclear Model of the Atom
The origins of the nuclear model of the atom can be traced to work done by Geiger, Marsden, and Rutherford from about 1909 to 1913. In 1911 Rutherford bombarded extremely thin metal foils with high energy alpha particles and found that most alpha particles went right on through these solid foils as though nothing were there. Less than one per cent of the alpha particles were deflected through small angles (<3º ) and a much smaller fraction (1:10,000) were deflected through large angles(>90º). Mathematical analysis showed that such a "scattering" pattern would occur if the mass of the atom were concentrated in a region about 10-4 Angstroms in diameter. Rutherford called this tiny massive object a "kernel". It was believed to be positively charged because the statistics of the experimental results were consistent with those calculated for coulombic scattering of the positively charged alpha particles.
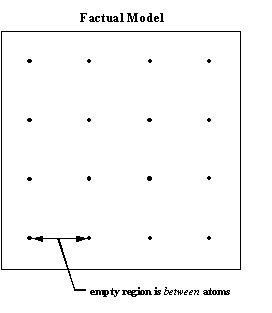
Ordinary macroscopic measurements and Avogadro’s number can be used to show that there is a volume associated with the atom, but it is on the order of one to five Angstroms in diameter or about 10,000 times the diameter of Rutherford’s kernel. The experiments showed that this volume is essentially empty. Illustrated on an everyday scale, an extremely thin metal foil would look something like a gigantic array of tiny dots, each dot about 1/10 millimeter in diameter (smaller than the periods at the ends of these sentences) and about a meter away from its neighbors. The whole array would be several football fields deep. If some similar dots were to be shot through this array, most dots would go right on through without hitting another dot or being deflected.
Prior to Rutherford’s experiment an aggregate of atoms was thought to be much like a collection of billiard balls in a shoebox. The balls were solid and impenetrable and were physically in contact with their neighbors. Unfortunately, scientists of Rutherford’s time interpreted the results of these experiments in terms of the already existing model. In other words, the balls were now hollow, like ping pong balls, and had a tiny, ultradense kernel in the center. This imagining of a sphere around each kernel had the effect of placing the emptiness of atomic aggregates within the atoms, rather than between the atoms. The atom thus acquired a "nucleus" and modern nuclear theory was born.
Factual and Conceptual Problems with the Nuclear Model
But as nuclear theory developed over the years, it was beset with serious conceptual problems that any highschool physics student will point out:
- Why don’t the protons violently repel one another, making the nucleus fly apart? How can neutrons, which have a half-life of 11 minutes, be part of a stable structure that lasts for billions of years? Why don’t the electrons neutralize the charge on the protons when placed in such extremely close proximity? Why don’t the electrons quickly radiate away their energy when moving so fast in such tight orbits? Why don’t they quickly spiral into the nucleus?
- Electrically neutral atoms both attract and repel one another, so why assume that the repulsion of the alpha particle originated from electrostatic forces? Couldn’t the charge just be extraneous? (note that the alpha particle is really a charged helium atom, not a subatomic particle like the neutron)
- A "nuclear force" was invented, ad hoc, to stick the parts of the nucleus together and make the neutrons stable. But there is no evidence that the nucleus has any parts to stick together in the first place!
- Atoms can be expected to have size and shape. Atoms with more mass should have larger size. An iron atom with 56 atomic mass units (a.m.u.) should be larger than a potassium atom of 39 a.m.u. But according to the nuclear model, the iron atom is smaller (7.1 ml/mole for iron versus 45.3 for potassium).
- Both graphite and diamond are made of carbon atoms. In diamond, the centers of the carbon atoms are separated by about 1.54 Angstroms in all three dimensions but in graphite, one of the dimensions changes to 3.4 Angstroms. The nuclear model thus implies that carbon atoms come in assorted sizes and shapes. But experience shows that for a given mass, all the atoms of a chemical element are identical. The variability is really between atoms, instead of within atoms, and can be altered by external influences like pressure and temperature.
The above claim that the nucleus is not made of parts requires some clarification. Atoms can be broken up into protons, neutrons and electrons. Therefore, scientists reason that these are "parts" of the atom and originate in the nucleus. But if I took a sledge hammer and smashed a color TV set all to pieces, have I revealed the actual "parts" by which a manufacturer assembles a TV set, or have I shown merely how a TV set disintegrates? The "atom smashing" experiments only show how the atom breaks up, not how it is put together.
Atom smashers blasted a whole zoo of supposedly "fundamental" particles out of the atom. Yet none of these could fundamentally be particles. Supposedly, an electron and a positron are both made of some combination of fundamental particles. Yet an electron and positron can be combined to produce two gamma rays (the so-called "annihilation" reaction). Conversely, two gamma rays can be combined to produce an electron and positron ("pair production"). But by definition, a truly fundamental particle cannot change into something else. Obviously, there is a suggestion of something fundamental here, but it is neither radiation nor particles. (It is also clear that spending $15 billion on a Super Conducting Super Collider would have been a complete waste of the taxpayer’s money.)
Scientists should have concluded that the thing that has all the mass of the atom and which accounts for all the properties of the atom, is, quite simply, the atom itself. Instead, they concluded it was a "nucleus" of the atom. Subsequently, they had to divorce the atom from known physical laws and the requirements of common sense. The result was a virtually incomprehensible model of the atom—one that is still being taught in our schools and colleges today.
Instead of concluding that we live in some kind of nightmare universe, physicists should discard the nuclear model of the atom and derive a fact-centered one that meets the requirements of common sense. Some hints on how to do this are presented in the table on the last page.
Science Corrects Itself Again and Again!
Historically, there is nothing at all unusual about erroneous theories being widely accepted for long periods of time by highly capable scientists. A classical example is the wide acceptance of the Ptolemic theories of the universe. Ptolemy, as you may recall, was a celebrated second century astronomer, geographer, and mathematician. His encyclopedic work, the Almagest was regarded as the ultimate authority on astronomy as late as 1543 A.D. (until Copernicus). He offered proofs that the earth was round, and explained the observed motions of the heavenly bodies with a very intricate combination of deferents, equants, epicycles, and circular orbits. His theories and their subsequent extensions and refinements had all the hallmarks of the truth: they reflected the best of scholarship, they explained all the observational data, and they had survived crucial tests for over a thousand years.
Yet his theory was fundamentally wrong. As is now established, the planetary orbits were heliocentric instead of geocentric and the orbits were elliptical instead of circular. There were no deferents or epicycles. As the telescope revealed, Ptolemy’s "divine beings" were not perfect or unchangable; the sun had spots and Venus had phases. Heavenly and earthly phenomena were of the same basic kind and man was not at the center of the universe. The whole theory collapsed not only because it was incredibly cumbersome and complex, but also because it was based on philosophical reasonings that were demolished by the accumulation of more precise observational data, and the availability of a much simpler explanation.
Can we make similarly huge mistakes today? Absolutely! Not only has human nature not changed, but we have more facts and interpretations available to us today than ever in history. We can make a bigger mess of things now than ever before! That is why scientists themselves warn us not to be complacent:
"History shows clearly that the advances of science have always been frustrated by the tyrannical influences of certain preconceived notions which were turned into unassailable dogmas. For that reason alone, every serious scientist should periodically make a profound re-examination of his basic principles." (New Perspectives in Physics, Louis de Broglie, 1962)
"It is proper to submit periodically to a very searching examination, principles that we have come to assume without any more discussion." (The Revolution in Physics, Louis de Broglie, (1953), p. 237)
"What men of one generation are pleased to regard as satisfactorily settled may be questioned by a succeeding one." (Understanding Physics Today, W.H. Watson, (1963), p. 7)
I hope articles such as this one will help people of science make that ‘profound re-examination of basic principles. (See also "Creativity in Science and Engineering", Ronald B. Standler,1998, http://www.rbs0.com/create.htm )
The Social Realizations
Discarding a major and widely accepted belief system, such as the nuclear model of the atom, will have important social consequences. A few realizations that will become more apparent are as follows:
1. Beliefs that even careful, ethical scientists hold to be thoroughly proven and completely beyond question can in fact be seriously wrong. Anyone who wants to become free of his own blind spots will have to work diligently to discover plausible alternatives to his own strongly held beliefs. As someone once said, "It is not what we don’t know that hurts us. It is what we do know that isn’t so."
2. Science is primarily a methodology based on factual data, repeatable experiments, and unequivocally testable hypotheses. Science is NOT what scientists believe. Reality is not derived from the beliefs of people. Experts can give us valuable insights, but they can also unintentionally mislead us.
3. Errors are often made at key, fundamental points. The large body of "knowledge" carefully derived from such erroneous foundations will also be faulty. Nevertheless, scientists can offer "convincing proofs" for everything beyond the foundation.
4. The truth is out there, but the people who know it may be hard to find, even though they are willing to be found.
5. The educational system, the popular press, and especially the scientific community, have not served adequately in describing factual information and in pointing out alternative interpretations of that information. What would happen to a university faculty member if he seriously proposed that the atom does not "have" a nucleus?
6. School boards that insist on teaching only "truth" must first decide what it is, and thereby remove the welcome mat for discussion of plausible alternatives. Truth is found by a great deal of persistent seeking and deep digging. It is not something that comes down out of heaven from God in a pop-top can, all prepared and pasteurized like a pre-canned belief system, ready to be consumed by ignorant students who are never supposed to question what they are consuming.
7. If requiring students to study "thoroughly proven" and "objective" science can give us cause for concern, we must also consider what side-effects the teaching of other subjects can have, such as the teaching of evolution and sex education. Likewise, requiring unthinking recitation of school pledges and prayers is more suited for "education of the masses" rather than education of individuals. Students need a sense of community and of participation, but the price must not be the needless destruction of individual values or individual initiative.
8. If it is possible to prove that the atom does not "have" a nucleus, then it may be entirely possible to do other things that are also thought to be impossible. The key to doing these impossible things rests more upon personal perception of primary facts rather than special problem-solving ability. One must also possess the ability to tolerate the mental chaos and uncertainty that occurs when questioning the validity of everything he thought he knew.
Undoubtedly the reader will be able to think of many others.
In this short presentation I must leave many questions both unasked and unanswered. I have neither the time nor the resources to write all that needs to be written on this topic. Instead, what I realistically hope to do is open some mental doors and appeal to "wealth in people" to finish the story.
Return to Scriptural Physics Home Page
Representations of a two-dimensional slice of an atomic lattice
The Nuclear Model |
The Atomic (factual) Model |
1. The atom is made of discrete particles: chiefly protons, neutrons, and electrons. The fundamental substance is intuitively incomprehensible. |
1. The atom is made of the Moseley substance. The Rydberg frequency and equations like E=cB and E=mc2 suggest that the fundamental substance is a ratio of space and time. The periodicity of the Periodic Table suggests that the internal structure of the atom is very simple and is based on squares of four integers: 1, 2, 3, 4. |
2. We know the atom is made of parts because it breaks up into all sorts of particles during atom smashing experiments. |
2. The atom smashing experiments show how the atom breaks up, not how it is put together. Ducks are not made of quacks just because quacks come out of ducks. Alpha particles, beta, and gamma rays are also common disintegration products, but physicists do not regard them as "parts" of the atom. |
3. Protons are parts of the nucleus. |
3. Protons would repel each other violently and so cannot be part of a stable structure like the ‘nucleus’. |
4. Neutrons are parts of the nucleus. |
4. Neutrons have a half-life of about 12 minutes and cannot be part of a stable structure lasting for billions of years. |
5. Electrons are parts of atoms. |
5. Electrons occur in matter but are not part of the atomic material. If electrons were orbiting a charged nucleus, they would quickly radiate away their energy and also neutralize the positive charge on the nucleus. |
6. A nuclear force holds the parts together and makes the protons, neutrons, and electrons stable. |
6. Because the atom is not made of parts, no nuclear "glue" is necessary. The nuclear force is just another assumption needed to hold the other assumptions together. |
7. The atom is made of some kind of fundamental particles. |
7. Both particles and radiation are interconvertible and hence neither can be truly fundamental. |
8. The size of the atom does not have a simple, regular relation to atomic weight. |
8. The size of the atom is directly proportional to the cube root of the mass number. |
9. The empty region in an aggregate of atoms is mostly within the atoms. |
9. The empty region in an aggregate of atoms is entirely between atoms. |
10. Atoms can have variable shapes and sizes as required, for example, in the diamond and graphite forms of carbon. |
10. Atomic size and shape do not vary. Interatomic spacing may vary under external influences like temperature and pressure. |
11. Aggregates of atoms are held together by electrical forces. |
11. Atoms are factually electrically neutral. Yet neutral atoms both attract and repel one another. Atoms in an ordinary iron bar, for example, strongly resist forces of tension and compression. |
12. Atoms exist only as abstract mathematical entities in multidimensional wave space. |
12. Atoms really do exist objectively. The nuclear model is unreal because the postulates used to construct it have been divorced from real physical laws. Nuclear theory says "the facts do not apply here." |
The atom is apparently composed of space/time ratios that take the form of intrinsic spin systems. For more on this theme see:
Inverseness, Complementarity, and the Wave/Particle Duality
Some Thoughts about Intrinsic Spin
Effects of Spin
The Origin of Intrinsic Spin
The Photon Spin System
The Atomic Spin System
One Christian's Perspective on Quantum Mechanics (a student paper)Books:
"Actually the electron is neither a particle nor a wave. It is a fundamental entity of matter, and it cannot be described by saying it is something else more familiar. . . . Likewise, the photon is neither a particle nor a wave. It also is a fundamental entity, characterized by certain properties."-- Introduction to Electromagnetic Fields and Waves, Charles, A Holt, 1963, p. 25
Prematurity in Scientific Discovery, edited by Ernest B. Hook, 2002
The Case Against the Nuclear Atom, Dewey B. Larson, 1963
"Resistance by scientists to scientific discovery", Bernhard Barber,
Science, 1961 Sep 1;134:596-602.Links:
"The Suppression of Inconvenient Facts in Physics", Rochus Börner, Ph.D., 2004.
http://www.suppressedscience.net/physics.htmlThe Reciprocal System, http://www.rsystem.org
The Collected works of Dewey B. Larson, http://www.rsystem.org/dbl/index.htmhtml 11/98g
Return to Scriptural Physics Home Page
Instructors, here is a riddle for your students:
Imagine three physicists standing in front of the class. Each makes a statement:
Physicist A: "The atom does not have a nucleus."
Physicist B: "A tiny little dot exploded and became the Universe as we know it."
Physicist C: "There are multiple universes with 11 dimensions. We inhabit only the "current incarnation" and only for an instant."
Then ask your students: '"Which of these statements is LEAST likely to be true?"
If the students answer B or C, then you can see that students are having trouble believing that modern physics is credible and relevant.
If they answer A, then draw a diagram on the board depicting nuclei with shells. Ask them to change only ONE concept. Suppose the "volume within the atom" is changed to be the "volume between atoms." Erase only the shells. Then ask them, What does that do to atomic theory? And does it affect your choice of A, B, or C? (The answer is not as important as is identifying the issues.)
Also, have them review the credibility criteria that NASA proposed for the Breakthrough Propulsion Physics program. ( www.bibliotecapleyades.net/ciencia/secret_projects/project301.htm )
See also:
http://10000solutions.org/solution/our-intuitions-are-wrong This URL has interesting comments. It is a project site for Arizona State University. Unfortunately, it typically has a lot of technical problems and may or may not be accessible.
Or see: Negative Knowledge .